
From: Comprehensive Analytical Chemistry, 2015. So greater the shielding effect, greater will be the size of an atom.\]įor helium (\(Z = 2\)), this gives \(-2.84765\) hartrees, an error of about \(2\%\) \((E_0 = -2.90372)\). Neutral metastable helium atoms or nitrogen molecules are extracted from the plasma and directed to a sample surface, where Penning ionization makes product ions that can be analyzed by mass spectrometry (MS) or IMS 26. So overall attractive force decreases and size increases. As the number of shells increases, the attraction between the nucleus and the outermost electron decreases or we can say the inner electrons shield the nucleus from valence electrons. The overall order of size of all these 3 species is as follows-Īpart from nuclear charge and number of shells there is another factor that decides the size of an atom which is the shielding effect. Thus the attractive force decreases and the size increases. The number of electrons increases but the number of protons remains the same. As with the hydrogen atom, the nuclei for multi-electron atoms are so much heavier. The origin of the coordinate system is fixed at the nucleus. The electrons are separated by a distance r12 r1 r2. It is formed when a hydrogen atom gains an electron. Figure 6.7.1 shows a schematic representation of a helium atom with two electrons whose coordinates are given by the vectors r1 and r2. The details about which combinations of protons and neutrons are stable and which are not is a very advanced topic and is not usually covered in General Chemistry. If there are too few or too many neutrons, the nucleus wont be stable. 5. Reason-Hydride $ \left( \right) $ is an anion of the hydrogen atom. But an atom must have just the right balance of protons to neutrons to make a stable nucleus.  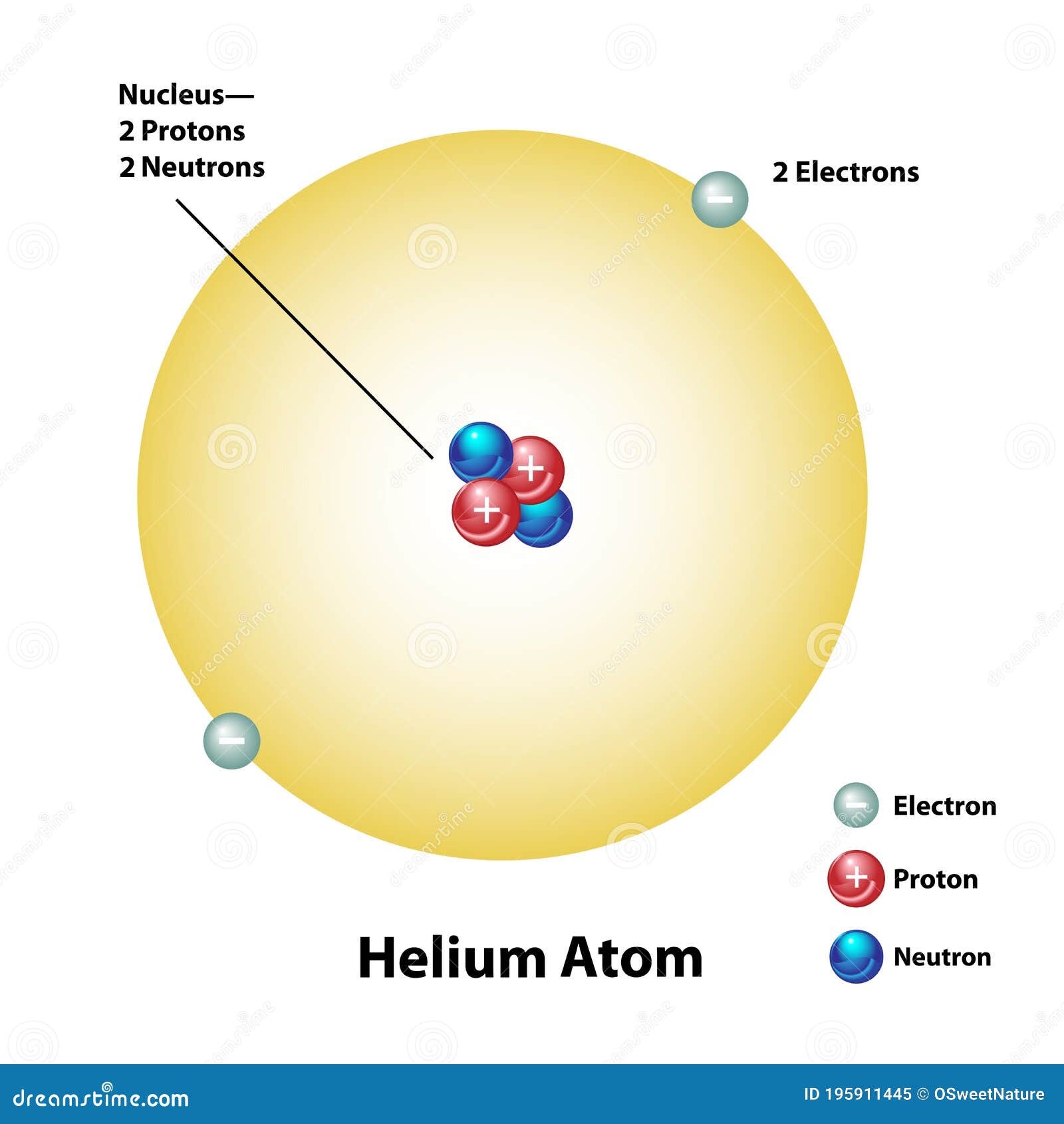
As discussed in Section 6.7, because of the electron-electron interactions, the Schrödingers Equation cannot be solved exactly for the helium atom or more complicated atomic or ionic species.However, the ground-state energy of the helium atom can be estimated using approximate methods. Using light species, especially helium, the scattering is predominantly. Variational Method Applied to the Helium Method. In another part of the question, we are asked why the hydride ion is larger than both hydrogen and helium atoms. Structural Information from Atomic Beam Diffraction K.-H. 
Its size is 31pm while the size of hydrogen is 53pm. The nucleus of the helium atom consists of two protons and two neutrons. Helium is the smallest element in the periodic table. Helium belongs to the right in the periodic table and as we move from hydrogen to helium number of inner shell electrons increases and effective nuclear charge increases and thus its size is smaller than the hydrogen atom. Reason-Hydrogen has 1 electron in its atomic shell while Helium has 2 electrons in its atomic shell. In the question, we are asked why helium is smaller than the hydrogen atom. The reason is as we move from top to bottom the number of atomic shells increases and thus the nucleus attracts the electron less so the effective nuclear charge decreases and thus increases atomic radius. Sources, facts, uses, scarcity (SRI), podcasts, alchemical symbols, videos and images. Depicted are the nucleus (pink) and the electron cloud distribution (black). (B)Atomic radius increases from top to bottom in a group of the periodic table. Element Helium (He), Group 18, Atomic Number 2, s-block, Mass 4.003. In the 1960s, theorists discovered that the fine-structure split (23P0-23P2) of the 23P energy level of helium is the best atomic system for measuring the fine structure constant (approximately. The reason is as we move from left to right the number of inner shell electrons increases and the nucleus attracts electrons more strongly and thus it’s effective nuclear charge increases which results in the decrease of atomic radius. Research objectives and content The aim of this project is to combine the advantages of helium surface scattering with recent developments in atom optics to. (A)Atomic radius decreases from left to right in a period of the periodic table. However when an atom gains or loses electrons it forms anions and cations respectively and their radius is called the ionic radius.īefore answering the question let us discuss the basic trends of the atomic radii. For a neutral gaseous atom, atomic and ionic radii are the same. It is the distance between the atomic nucleus and the outermost valence shell electron. Atomic radius is used to measure the size of an atom. Hint: To answer the question we need to know the concept of atomic and ionic radius.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
